Our Services

Regulatory Affairs & Registration
We act as a direct interface between manufacturers, distributors, and the Egyptian Drug Authority (EDA), supporting products throughout their regulatory journey.
- Medical device classification according to Egyptian regulations
- Preparation, review, and submission of registration files
- Handling regulatory queries and follow-up with authorities
- Support for importation approvals and regulatory logistics
- Assistance with renewals, variations, and updates
- Post-market compliance and vigilance support
Our approach focuses on accuracy, completeness, and proactive follow-up, reducing delays and avoiding unnecessary regulatory obstacles.

Marketing Strategy & Scientific Support
We help manufacturers translate regulatory approval into real market presence.
- Market entry and product launch planning
- Market and competitor analysis
- Scientific positioning and value messaging
- Support for ethical and compliant promotional activities
- Coordination of educational and awareness initiatives
All marketing support is aligned with local regulations, professional ethics, and healthcare standards, ensuring credibility and sustainability.
Distributor Follow-Up & Market Support
Choosing the right distributor is critical. MedOut supports manufacturers before and after distributor appointment.
- Distributor search, evaluation, and selection
- Verification of licenses and regulatory readiness
- Support in defining roles, responsibilities, and KPIs
- Ongoing follow-up on performance, stock, and reporting
- Training distributor teams on regulatory and compliance expectations
This ensures regulatory alignment, operational stability, and long-term market growth.
Training & Knowledge Development
We provide professional training programs tailored to the medical device sector, including
- Regulatory Affairs fundamentals
- Documentation and compliance best practices
- Market access and distribution readiness
- Communication and professional skills
Our training is practical, experience-based, and designed to empower teams working within the Egyptian market.
Our Product Categories
Medical Devices
Capital equipment, surgical instruments, and consumables.
In-Vitro Diagnostic (IVD) Kits
Reagents, calibrators, and diagnostic kits for clinical use.
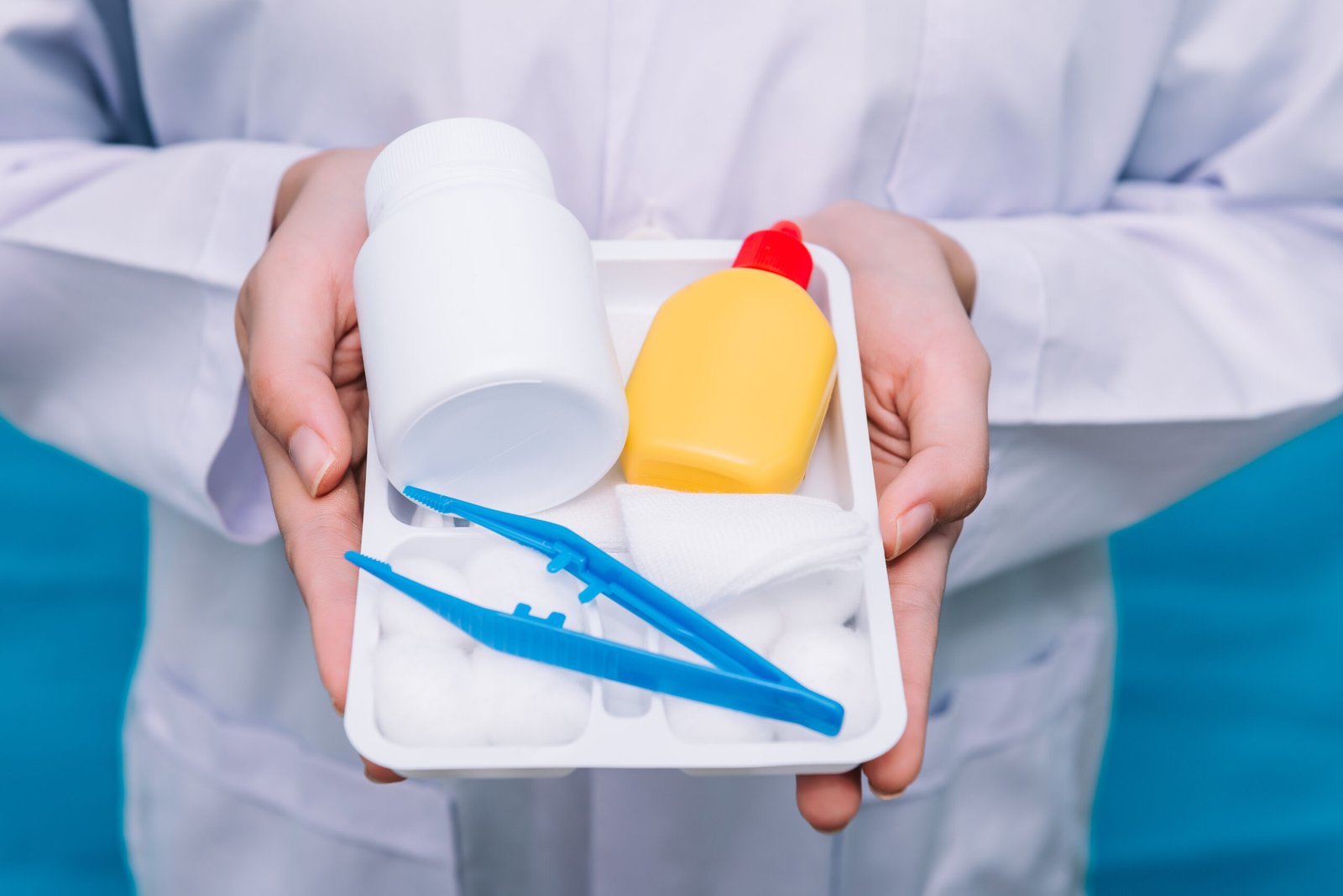
Specialized Medical Kits
Procedure-specific kits and sets that meets requirements and purpose
Not sure what you need ?
Tell us about your products and current stage—entry, registration, portfolio maintenance, or distributor performance—and we’ll propose the best path forward.
Contact Us
Phone
+20 2 2364 0802
info@medout-eg.com
Website
www.medout-eg.com
Address
28 Al Mekyas, Manial Al Rawda, Cairo, Egypt
